Exactech Recalls Connexion GXL Hip Liner and Knee & Ankle Replacement Parts
The Exactech medical implant attorneys at Hollis, Wright & Clay, P.C. are currently handling claims on behalf of injured individuals throughout the entire United States.
Exactech, a global medical device company, develops and markets orthopedic implant devices. Exactech recently recalled products related to both hip replacement surgery and knee and ankle surgeries. The two products are: the Connexion GXL Hip Liner and the Ultra-High Molecular Weight Polyethylene (UHMWPE) Knee and Ankle Polyethylene Inserts.
If you or a loved one has been injured by a hip, ankle or knee implant manufactured by Exactech, contact the experienced Exactech medical implant injury attorneys at Hollis, Wright & Clay, P.C. here or at 844.LAW.TALK or 205.324.3600. We will fight for you to ensure you are compensated for medical bills, lost wages, pain and suffering, emotional distress, permanent injury and more.
Exactech Connexion GXL Hip Liner What is the Exactech Connexion GXL Hip Liner?
What is the Exactech Connexion GXL Hip Liner?
The liner is intended to be used by surgeons during a hip replacement surgery and is placed into the patient’s hip socket (acetabular cup) and, according to the manufacturer, should be less prone to wear and tear due to the radiation process used to create it. The product is made of a plastic called polyethylene.
What is happening to the Exactech Connexion GXL Hip Liner?
While the product was created and intended to reduce wear and tear, it is actually accelerating wear and tear. The liners can deteriorate earlier than expected and unfortunately, can result in the patient having to undergo a revision surgery. Reports indicate that the early wear and tear may be attributed to significant flaws in the design of the liner.
Recently, surgeons from the Hospital for Special Surgery in New York warned of significant wear of the liner shortly after surgery. Additionally, the surgeons noted bone deterioration within 5 years of surgery. The surgeons described the design defect in the liner as “catastrophic” and recommended the medical community monitor very carefully patients with the Exactech Connexion GXL Hip Liner.
What can occur if your device is failing?
If you’ve had a hip replacement surgery in which the surgeon used the Exactech Connexion GXL Hip Liner and are experiencing any of the following, contact us here or at 844.LAW.TALK.
- Pain,
- Stiffness,
- Trouble moving,
- Joint weakness,
- Bone degeneration (Osteolysis), and
- Revision surgery.
The Exactech medical implant attorneys at Hollis, Wright & Clay, P.C. urge anyone who has any of these symptoms, or especially is going to need a revision surgery due to complications from the liner, to contact us immediately. You are likely entitled to compensation for medical bills, lost wages, pain and suffering and emotional distress. Don’t take on large medical device companies on your own. Call us at 844.LAW.TALK or 205.324.3600 or contact us online here.
Exactech Ultra-High Molecular Weight Polyethylene (UHMWPE) Knee and Ankle Polyethylene Inserts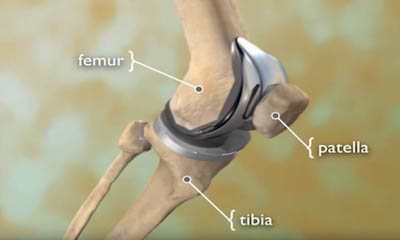 What is the Exactech UHMWPE Knee and Ankle Insert?
What is the Exactech UHMWPE Knee and Ankle Insert?
The Exactech plastic inserts are used in knee and ankle replacement surgeries. The product fits in between metal components in knee and ankle replacements to cushion joints. Exactech in 2021 issued a recall for thousands of plastic inserts because the plastic may wear early, leading to implant failure and revision surgery.
The manufacturer recalled the device because the product’s packaging didn’t adequately protect the inserts from oxygen exposure. This exposure decreases the effectiveness of the product.
What is wrong with the insert?
After the initial recall in 2021, Exactech issued an updated recall letter to surgeons throughout the United States, detailing information about more than 100,000 defective and dangerous inserts implanted in patients since 2004.
Any patient who received the now-recalled knee or ankle insert may experience problems that could result in a revision surgery. A knee replacement system includes several components, including the femoral implant, tibial tray, patellar component, and a tibial polyethylene insert. The problem with the Exactech product is in the polyethylene insert, which serves an important role in the knee functionality and longevity by functioning as cartilage in the knee joint.
Problems that are associated with early wear of Exactech’s UHMWPE Knee and Ankle Insert
If you experience any of the below symptoms and have received an Exactech UHMWPE Knee and Ankle Insert, we urge you to contact us immediately.
- Debris production,
- Bone loss,
- Component fatigue,
- Component cracking and,
- Revision surgery.
Patients with these issues may begin suffering from new or worsening swelling, pain, inability to bear weight, instability, loosening, clicking, and more.
Broadspire Claims ProcessAccording to Exactech, it has contracted with a company known as Broadspire to handle claims made by patients that received one of these recalled implants. Broadspire will obtain your personal information, product identification information and will also ask for information related to your out-of-pocket expenses and damages. The Exactech medical implant attorneys at Hollis, Wright & Clay, P.C. recommend that patients do NOT go through the Broadspire claims process without having their claim evaluated by an attorney. It is likely that if you receive money from Exactech through the Broadspire claims process, you will be required to sign a settlement and release agreement that will forever release and discharge Exactech from any further liability. Additionally, it is likely that the Broadspire claims process will result in the patient being grossly undercompensated for their injuries.
Exactech Attorneys at Hollis, Wright & Clay, P.C. are Here for YouOur attorneys are standing by to help you with your defective Exactech UHMWPE Knee and Ankle Insert or your Exactech Connexion GXL Hip Liner. All cases are handled on a contingency fee basis which means we don’t get paid unless we successfully recover money on your behalf. Contact us online here or call us at 844.LAW.TALK or 205.324.3600. Consultations are free.
 Hollis Wright Home
Hollis Wright Home
